Projects
Check out the Research Projects sponsored by the COBRE Center:

Roberto Rodríguez, Ph.D.
- Instagram de su laboratorio: @brainsinthedark
- ORCID ID: 0000-0002-7716-1342
- LinkedIn: https://www.linkedin.com/in/roberto-rodriguez-morales/
Biography
Dr. Roberto Rodríguez is an Assistant Professor in the Department of Anatomy & Neurobiology at the University of Puerto Rico (UPR) School of Medicine. His research explores the mechanisms underlying social behavior adaptation using fish models. He earned his PhD in Anatomy at the UPR School of Medicine, where he investigated genetic mechanisms of sensory system regeneration in zebrafish, applying advanced tools such as CRISPR/Cas9 genome editing.
As a postdoctoral fellow at Lehigh University under the mentorship of Dr. Johanna Kowalko, Dr. Rodríguez examined the repeated evolution of aggressive behavior in the blind Mexican cavefish, Astyanax mexicanus. His contributions as a trainee have been published in journals including Zebrafish, Gene Expression Patterns, and BMC Ecology and Evolution.
In 2023, Dr. Rodríguez returned to UPR to establish his independent laboratory, focused on the intersection of sensory system function and social behavior in A. mexicanus. His program has been supported by multiple awards, including pilot project grants from the RCMI and COBRE Neuroplasticity Center at UPR, as well as an NIH R16 SuRE-First award from the National Eye Institute. He now also leads a research project within the Puerto Rico Center for Microbiome Sciences (COBRE Phase I), investigating how gut microbiome diversity shapes social behavior in cavefish. His mentor is Dr Riccardo Papa a Professor of Evolution at UPR Rio Piedras.
Keywords
- Mechanisms underlying sensory system adaptation (microbiota diversity, genetic diversity, etc.)
- Sensory system plasticity (lateral line)
- Sensory system regeneration (lateral line)
Project summary
Social behaviors are a hallmark of life across the animal kingdom, from insects like flies and ants to vertebrates such as fish, amphibians, rodents, and humans. Yet, the display of these behaviors varies widely: some species live in complex societies, while others are solitary. Even within the same species, populations may differ in how social they are. A striking example is the Mexican tetra (Astyanax mexicanus), which exists as two forms—highly social, river-dwelling surface fish and blind, cave-dwelling fish that have largely lost social behaviors. Our lab is interested in uncovering the biological mechanisms that drive this repeated loss of sociality in cavefish populations.
One of our central projects, supported by the Puerto Rico Center for Microbiome Sciences, investigates how gut microbiome diversity influences social behavior adaptation. Using a resident–intruder assay, we focus on aggression and social hierarchy formation to address two key questions: (1) Do dietary changes or microbiome imbalances alter social dynamics in surface and cavefish? (2) Are specific microbiome signatures linked to dominance, subordination, or aggression? This work will clarify how the gut microbiome contributes to both social and asocial phenotypes in a unique natural model.
In parallel, we study how blind cavefish have adapted through expansions of the mechanosensory lateral line—a superficial system of hair cells similar to those of the vertebrate inner ear. By investigating the developmental mechanisms that drive these expansions, we aim to identify genetic factors that promote sensory growth and test whether such compensations also influence social behavior.

Fig. 1. The Mexican tetra, Astyanax mexicanus is a vertebrate fish that exists as two morphotypes: ancestral surface fish (top), and at least 30 populations of cave-adapted, blind cavefish (bottom).
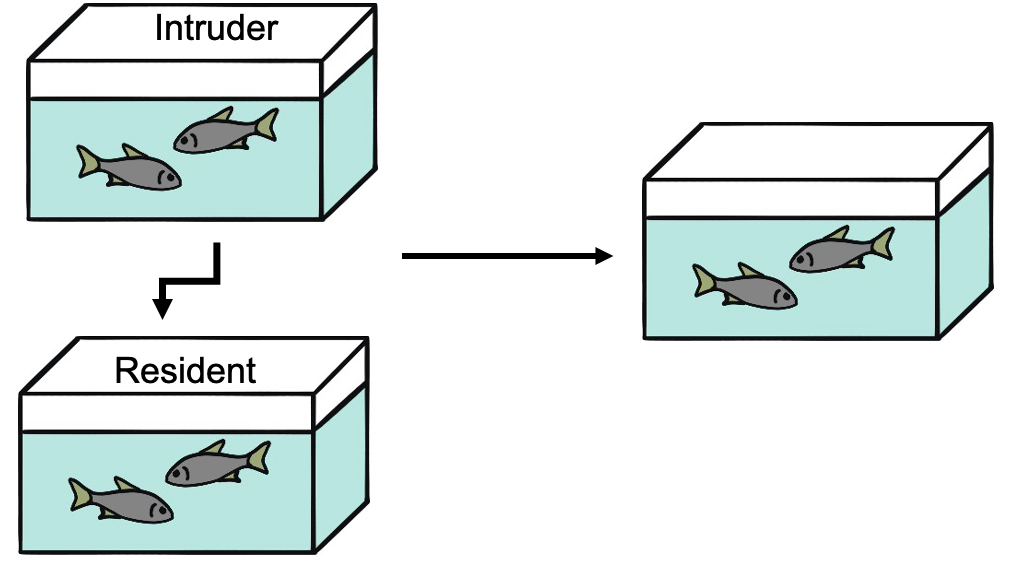
Fig. 2. The resident-intruder assay is used to quantify aggressive interactions in fish dyads of the Mexican tetra.
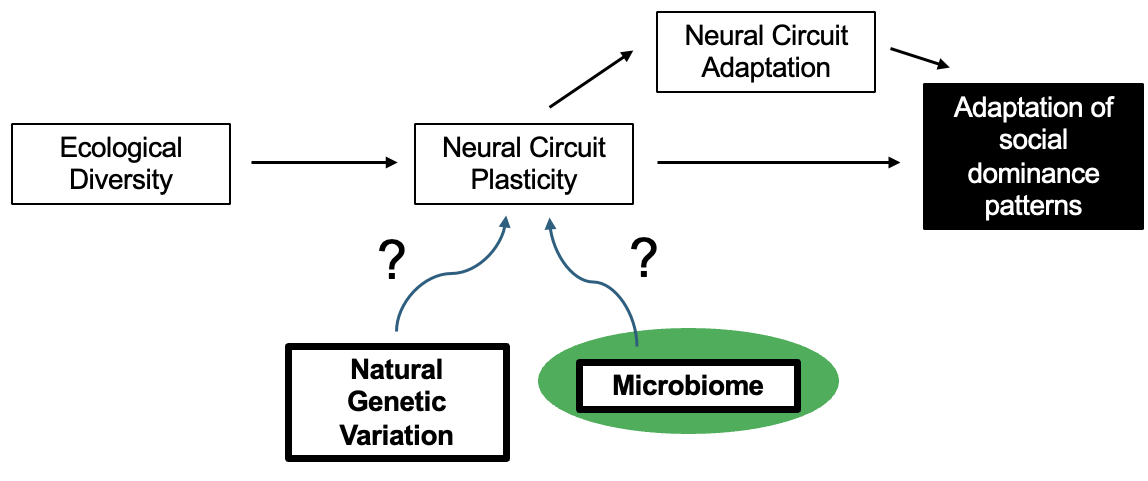
Fig. 3. The mechanisms underlying social behavior adaptation are multifactorial, including natural genetic variation, and potentially gut microbiome diversity.

Meet Roberto’s Team:
Fabiola Pagán-Torres –
Alana del Coral López-Cruz –
Dhalma Bayron-Ho –
Julián Navarro-Pagán –
Angel Márquez-Otero –
Paola Figueroa –
Alexander Figueroa –
Luis Perez –
Sofia Cuevas –
Jamilette Crespo –
Carlos Gonzalez –
Homar Cardona –
Adrian Rivera –
Axel Rodriguez –

Yancy Ferrer, Ph.D.
Assistant Professor
Dept Anatomy and Neurobiology, UPR School of Medicine
- Instagram:
- ORCID ID: https://orcid.org/0000-0001-6489-3272
- LinkedIn: https://www.linkedin.com/in/yancy-ferrer-acosta-7b795667/
- Google Scholar: https://scholar.google.com/scholar?hl=en&as_sdt=2007&q=%22Yancy+Ferrer-Acosta%22&btnG=
-ResearchGate: https://www.researchgate.net/profile/Yancy-Ferrer?ev=hdr_xprf
Biography:
Dr. Yancy Ferrer-Acosta is an Assistant Professor and researcher whose career integrates neuroscience, cancer biology, and translational science. She earned his B.S. in Biology from the University of Puerto Rico (UPR)–Río Piedras, followed by a Ph.D. in Biology (specialty in Neuroscience) in the same Campus, where she investigated mechanisms of tau aggregation in neurodegenerative diseases. She then pursued postdoctoral training at the UPR, Rio Piedras Campus and the MSRC, focusing on the field of cancer and immunology.
Dr. Ferrer has built a research program centered on neuroprotection and lung cancer therapies. Her work explores nanoparticle-based drug delivery across the blood–brain barrier in an in vivo epilepsy model, natural neuroprotective compounds such as tobacco-derived cembranoids in stroke, and the role of immune receptors in neuroinflammation. In parallel, she investigates therapeutic strategies for non-small cell lung carcinoma, including the use of natural compounds and targeted nanotechnology-based approaches.
With extensive teaching and training experience, Dr. Ferrer is committed to mentoring the next generation of biomedical scientists. She has received research funding, authored peer-reviewed publications, and actively participates in national and local scientific meetings. Her long-term goal is to translate laboratory findings into innovative therapies for neurological disorders and cancer.
Keywords: Neuroprotection and cancer therapeutics using natural products and nanotechnology
Project summary
Current antiepileptic drugs are often ineffective for about 30% of patients and cause significant side effects, underscoring the need for new treatments. Recent research links gut dysbiosis to epilepsy, showing that disrupting the gut microbiome increases seizure susceptibility and affects cognition and behavior. Positive outcomes have been observed in epilepsy patients by altering the gut microbiota through probiotics, diet changes, or fecal matter transplants. Gut microbiota may protect against epilepsy by modulating neuroimmune interactions and reducing neuroinflammation. Probiotic treatments have shown promise in increasing GABA and neurotrophic factors, reducing inflammatory cytokines, and decreasing seizures. However, the impact of treatments on the epileptic gut microbiome remains poorly understood.
Our study, in collaboration with Dr. Yamixa Delgado’s laboratory aims to address this gap by testing Edelfosine (Ef), an FDA-approved investigational anti-neoplastic drug and HIV-1 latency reversal agent, as a novel epilepsy treatment. Our research reveals that the synthetic lysophospholipid edelfosine (Ef), delivered via a brain-targeted drug delivery system (Tf-SA-Ef-DDS), is an anti-inflammatory molecule and an effective anti-convulsant in a pilocarpine-induced epilepsy mouse model. This project explores whether Ef impacts the gut microbiota and metabolites following an epileptic insult. We will do this by collecting fecal matter of naïve animals and epilepsy-induced animals before and after the seizure and analyzing their gut microbiota, its metabolites, and brain metabolites. We will compare the obtained data in Ef-treated and untreated mice. We hypothesize that Ef will promote an anti-inflammatory microenvironment within the gut by protecting the survival of butyrate-producing taxa such as Bifidobacteria and Lactobacillus after an epileptic insult.
The Center for Microbiome Studies (CMS) 16S sequencing and metabolomic cores, together with the mentoring of Dr. Godoy and microbiome experts, our lab will be able to delve into this new and exciting field, in our search for medications against epilepsy, that can treat brain and gut.
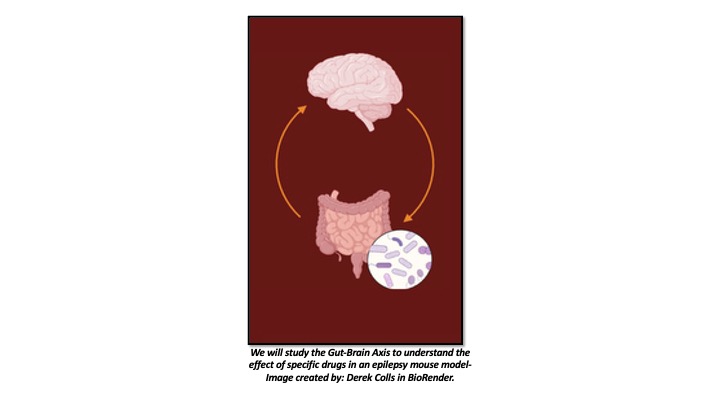
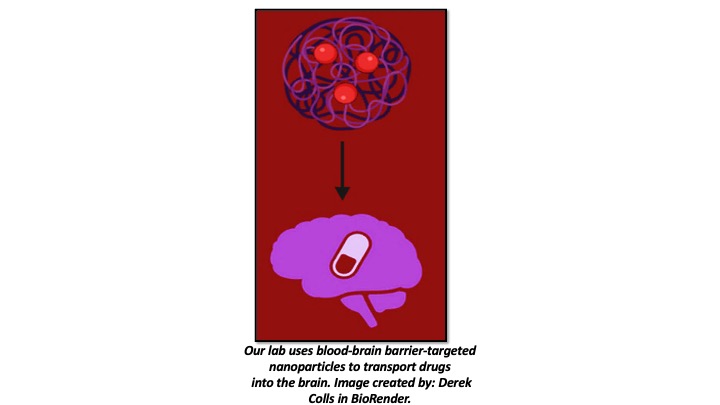
Other Research Interests
Our laboratory focuses on advancing research in neuroprotection and lung cancer through both synthetic and natural therapeutic strategies. In the area of neuroprotection, we investigate:
(1) Smart nanoparticles designed to cross the blood–brain barrier and deliver protective drugs, tested in epilepsy models and in vitro systems, and their effect on the gut microbiome.
(2) Tobacco-derived cembranoids that naturally penetrate the blood–brain barrier and show promise in reducing stroke damage, explored through rat stroke models and mechanistic studies.
(3) The role of the immunoreceptor TREM-Like Transcript-1 (TLT-1) in brain cells following neuroinflammation, by using immunofluorescence, histology, and molecular biology approaches.
In our lung cancer projects, we employ the Lewis Lung Carcinoma model to evaluate therapeutic strategies against non-small cell lung carcinoma (NSCLC), including the use of (1) natural products such as cembranoids to inhibit tumor growth, and (2) the development of targeted, biocompatible nanoparticles to deliver anti-tumor drugs.
Collectively, our work seeks to develop innovative therapies for neurological disorders and lung cancer, emphasizing natural compounds, nanotechnology, and immune-related mechanisms.
Link: https://md.rcm.upr.edu/anatomyneurobiology/dt_team/yancy-ferrer-phd/
Meet the Ferrer Lab Team

Banner prepared by my students
- José G. Cirino Simonet-
This email address is being protected from spambots. You need JavaScript enabled to view it. - 6th year graduate student completing his Ph.D. in Cellular and Molecular Biology, Universidad Central del Caribe, School of Medicine, Bayamon - Kelvin O. Rodríguez Rivera-
This email address is being protected from spambots. You need JavaScript enabled to view it. - 2nd year Graduate student completing his Ph.D. in Anatomy and Neurobiology, UPR-MSC - Derek J. Colls García-
This email address is being protected from spambots. You need JavaScript enabled to view it. - 6th Year Undergraduate student at UPR-Rio Piedras (will enter the lab in January as graduate, Ph.D. Intercampus student) - Alexander Montalvo Berríos-
This email address is being protected from spambots. You need JavaScript enabled to view it. - 4th year Undergraduate student UPR Bayamon - Paola N. Gracia Ayala-
This email address is being protected from spambots. You need JavaScript enabled to view it. - 3rd year Undergraduate student-UPR-Bayamón - Arot L. Velázquez Pulliza-
This email address is being protected from spambots. You need JavaScript enabled to view it. 3rd year Undergraduate student at UPR Rio Piedras - Abriel Rivera Rivera-
This email address is being protected from spambots. You need JavaScript enabled to view it. 2nd year Undergraduate student-Interamerican University, Arecibo

Stephanie Dorta-Estremera, Ph.D.
Associate Professor
Dept. Microbiology and Immunology, UPR School of Medicine
- Instagram: https://www.instagram.com/dortalab?igsh=OWx3YXVsZjBxaXFz
- ORCID ID: https://orcid.org/0000-0003-4440-6806
- LinkedIn: https://www.linkedin.com/in/stephanie-dorta-estremera-1a621a87/
- Google Scholar: https://scholar.google.com/citations?hl=en&tzom=240&user=lvzur3QAAAAJ
- Página web de su laboratorio
https://md.rcm.upr.edu/micro/dt_team/dr-stephanie-m-dorta/
Biography
Dr. Dorta-Estremera is a basic and translational immunology researcher who has focused her career on characterizing immune mechanisms modulating the pathogenesis of different diseases such as HIV, autoimmune diseases, and cancer, as well as treatment responses for these diseases. She obtained her B.S. in Biology at the University of Puerto Rico, Rio Piedras Campus. She did a post-baccalaureate training at the National Institutes of Health and obtained a Ph.D. in Immunology from the University of Texas Health Sciences Center MD Anderson Cancer Center. Her doctoral degree focused on characterizing the immune-mediated mechanisms involved in the development of lupus. Her postdoctoral training at MD Anderson Cancer Center focused on cancer immunology research and identifying immune correlates for the protection of novel immunotherapies and for chemoradiation in mouse preclinical models and cancer patients. Now, as an assistant professor at the UPR-Medical Sciences Campus and UPR Comprehensive Cancer Center, she is developing a research program to dissect immunological mechanisms involved in treatment resistance on HPV-related cancers, to understand the interaction between microbiota and immune cells in mucosal tissues, and to identify immune and microbial biomarkers that could be targeted to improve current therapies, especially in the Puerto Rican population.
Keywords
- Cancer Immunology
- HPV-related cancers
- Microbiome and host interactions
- Immune monitoring by flow cytometry
Project summary
Patients with oropharyngeal cancer (a type of throat cancer) are often prescribed antibiotics to prevent infections after surgery; however, antibiotics can reduce the diversity of good bacteria in the gut and the oral cavity, which may weaken the immune system and lower the effectiveness of cancer treatments like chemotherapy and immunotherapy.
Some studies show that antibiotic use increases the risk of death in patients with head and neck cancers who receive immune checkpoint therapy. However, not all antibiotics act the same way, posing the question of which antibiotics may be better or worse in terms of their impact on the oral and gut microbiota, immune responses, and response to cancer therapies in oropharyngeal cancer. Therefore, we hypothesize that different antibiotics decrease gut and oral microbial diversity and differentially impact anti-tumor immune responses and oropharyngeal cancer development.
This research project has two goals:
- Test antibiotics in mouse models of oropharyngeal cancer to see how oral and topical (on the tongue) antibiotics affect tumor growth, immune responses, and cancer treatment responses.
- Study the effects of antibiotics on oral bacteria and inflammation in Hispanics at high risk for cancer, to see how antibiotics change oral bacteria and inflammatory markers related to oral cancer risk.
The long-term goal is to understand which antibiotics harm or help cancer patients so doctors can make better treatment choices and improve survival.
Other projects in the laboratory:
- Identification of immune and microbial biomarkers associated with cancer treatment responses
- Díaz-Rivera J, Rodríguez-Rivera MA, Meléndez-Vázquez NM, Godoy-Vitorino F, Dorta-Estremera SM. Immune and Microbial Signatures Associated with PD-1 Blockade Sensitivity in a Preclinical Model for HPV+ Oropharyngeal Cancer. Cancers (Basel). 2024 May 30;16(11):2065. doi: 10.3390/cancers16112065. PMID: 38893183; PMCID: PMC11171047.
- Colbert LE, El Alam MB, Wang R, Karpinets T, Lo D, Lynn EJ, Harris TA, Elnaggar JH, Yoshida-Court K, Tomasic K, Bronk JK, Sammouri J, Yanamandra AV, Olvera AV, Carlin LG, Sims T, Delgado Medrano AY, Napravnik TC, O'Hara M, Lin D, Abana CO, Li HX, Eifel PJ, Jhingran A, Joyner M, Lin L, Ramondetta LM, Futreal AM, Schmeler KM, Mathew G, Dorta-Estremera S, Zhang J, Wu X, Ajami NJ, Wong M, Taniguchi C, Petrosino JF, Sastry KJ, Okhuysen PC, Martinez SA, Tan L, Mahmud I, Lorenzi PL, Wargo JA, Klopp AH. Tumor-resident Lactobacillus iners confer chemoradiation resistance through lactate-induced metabolic rewiring. Cancer Cell. 2023 Nov 13;41(11):1945-1962.e11. doi: 10.1016/j.ccell.2023.09.012. Epub 2023 Oct 19. PMID: 37863066; PMCID: PMC10841640.
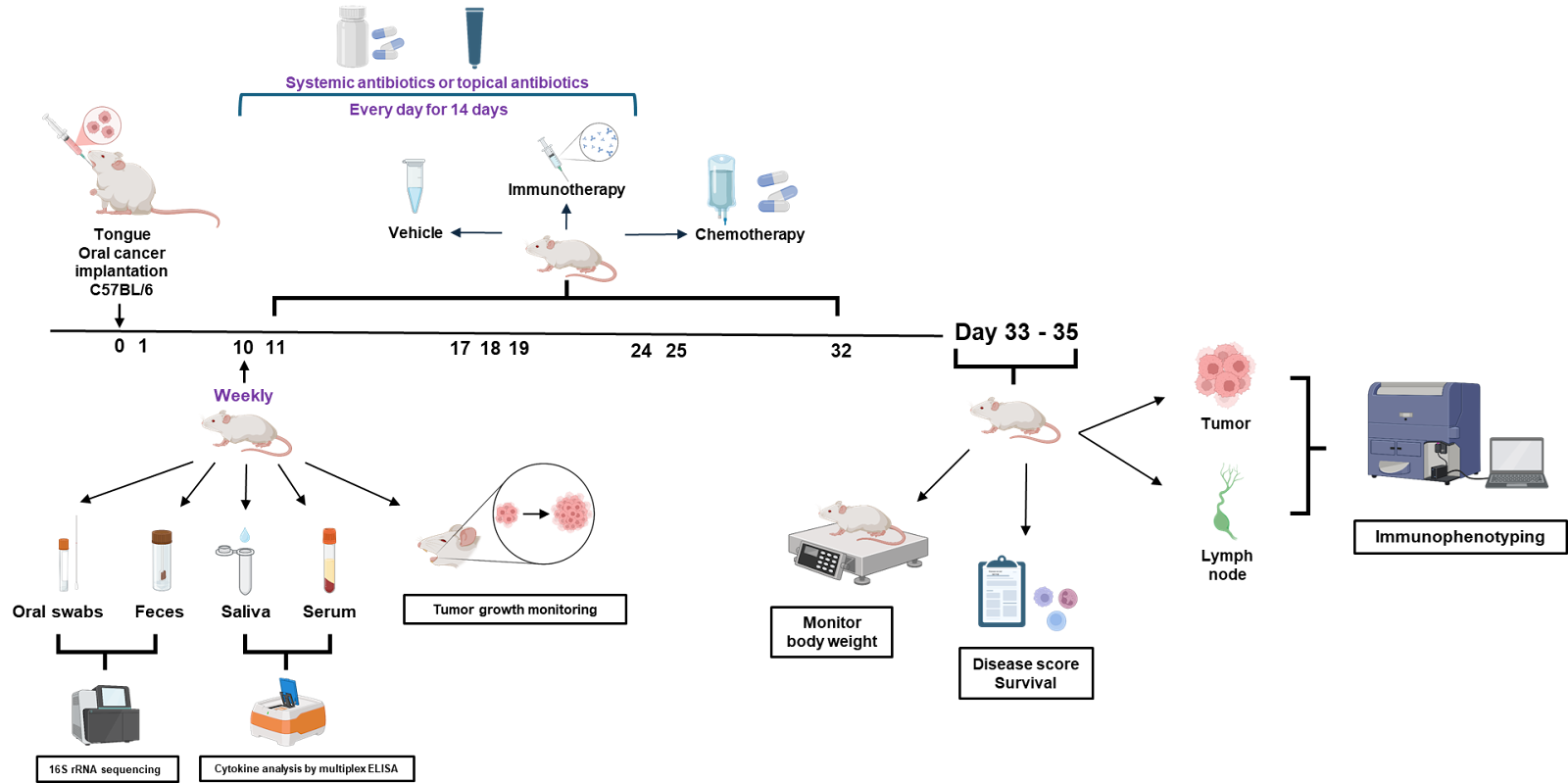
Figure 1. In this aim, we will determine the biological effect of oral and topical antibioticson cancer treatment responses in preclinical models of oropharyngeal cancer
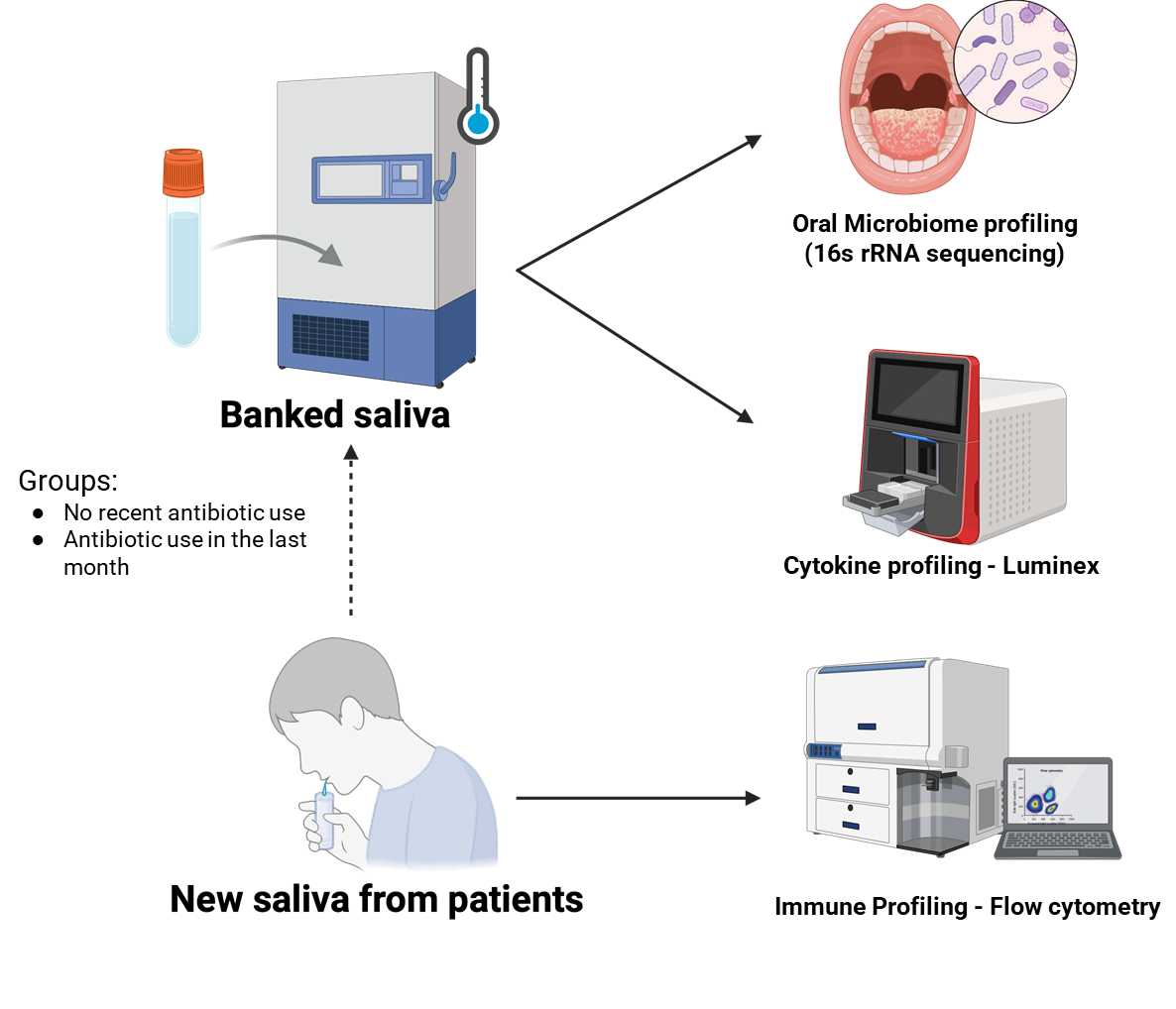
Figure 2. In this aim, we will identify the impact of antibiotic use on the oral microbiota and inflamation in individuals with a high risk of developing cancer
Meet Stephanie’s team:

Michael Rodríguez Rivera –
Aryanne Torres Ventura –
Liz Velázquez Rivera –
Andy González Hernández –
Emmanuel Colón Rodríguez –
Nicolle Colón Ortiz –
Isabel Colón –
Joshua Meléndez Calderon –
Mónica Cintrón Santiago -